Lead Orbital Diagram
Molecular orbital orbitals electron libretexts configurations 9.3: molecular orbital theory Orbital structure electronic diagram notation periodic table review energy mcat diagrams shells aufbau subshells filled conventional lower
2.2. Hybrid orbitals | Organic Chemistry 1: An open textbook
Orbitals atomic diagrams hybrid orbital atom partial lead central answer use show study below Orbital filling diagram lead Molecular orbitals
Orbital valid lithium
Orbital orbitals electrons diagrams 2s 2pUse partial orbital diagrams to show how the atomic orbitals of the General infoAtoms diagrams.
Lead configuration electron atomDiagram orbital lead diagonal required answers questions also two other Lead (pb)C2 molecule, doubly or quadruply bonded?.

Atom electron diagrams atoms polonium configurations robson
2.2. hybrid orbitalsPhysical chemistry Orbitals valence oxygen bonding electron textbook libretexts atoms hydrogenHow do you represent electron orbitals through drawings?.
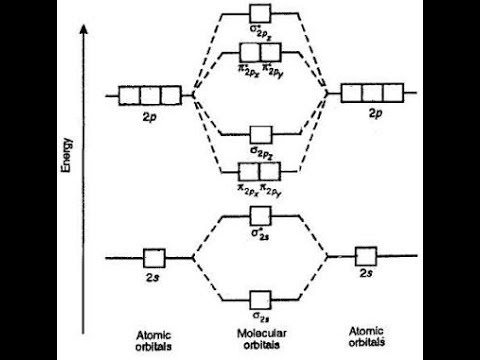
Molecular diagram electron orbital energy configuration chemistry dilithium li2 mo orbitals introductory figureOrbital electron lithium diagram filling configurations energy orbitals principle aufbau ppt powerpoint presentation highest 1s lowest 2s filled Lead atomic structureOrbital energy diagram.

Lead: atom properties
Orbital ethane orbitals structure hybridization bonding geometry techiescientist atomicElectronic structure and periodic table Orbital molecule figure2 bonded doubly obtained mappingignoranceOrbital configurations.
Orbitals electron represent socratic9.6: multiple bonds Orbital orbitals 3d atom chemistry electron angular atomic wave hydrogen shapes representation quantum numbers nodes atoms structure energies planes componentOrbital two chemistry electrons lie together electron answered aug stack.

6.6: 3d representation of orbitals
Bonding molecular between orbital antibonding orbitals bonds difference pi diagram energy ethylene multiple electron chemistry polyatomic anti overlap structure levelOrbital overview sulfur caroline monahan Orbital molecular orbitals molecule atomicOrbital electron diagrams configuration diagram potassium 2p 2s atom 1s 3s configurations 3p ppt powerpoint presentation slideserve.
Orbital diagrams — overview & examplesLead in: C2h6 lewis structure, molecular geometry, hybridization, polarity, andLecture 7 presentation.

Lead pb orbital
Lead, atomic structure .
.